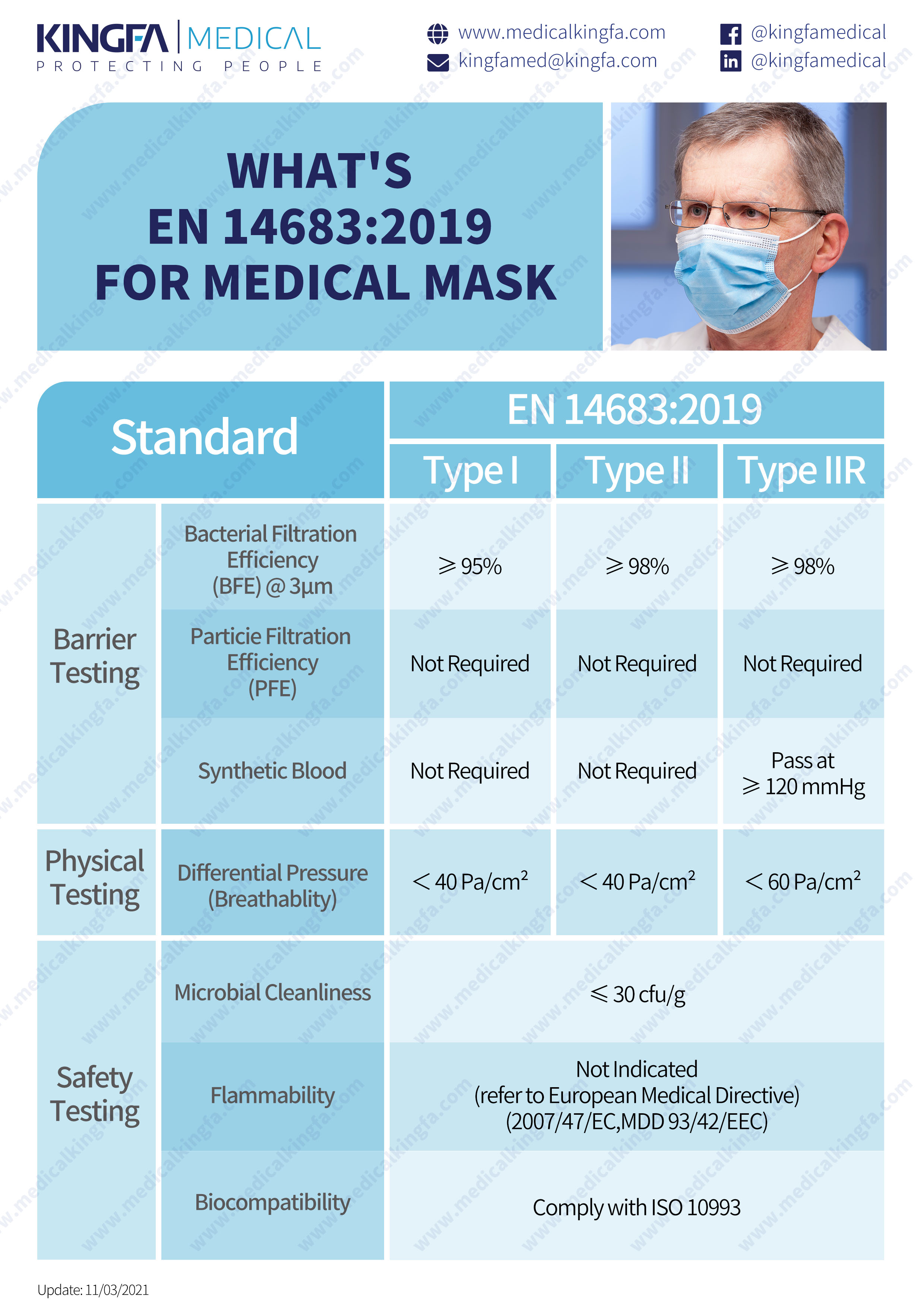
Type I
Type I medical face masks should only be used for patients and other persons to reduce the risk of spread of infections particularly in epidemic or pandemic situations. Type I masks are not intended for use by healthcare professionals in an operating room or in other medical settings with similar requirements.
Type II
Type II mask (EN14683) is a medical mask minimize the direct transmission of infective agent between staff and patients during surgical procedures and other medical settings with similar requirements. Type II masks are principally intended for use by healthcare professionals in an operating room or other medical settings with similar requirements.
Type IIR
IIR mask EN14683 is a medical mask to protect the wearer against splashes of potentially contaminated liquids.. The IIR mask includes a splash-proof layer to prevent blood and other body fluids from entering. IIR masks are tested in the exhalation direction (from inside to outside), taking into account the efficiency of bacterial filtration.
What is the difference between type I and type II masks?
The BFE (bacterial filtration efficiency) of type I mask is 95%, while the BFE of type II and II R masks are 98%. The same breathing resistance of type I and II, 40Pa. dical face masks specified in this European Standard are classified into two types (Type I and Type II) according to bacterial filtration efficiency whereby Type II is further divided according to whether or not the mask is splash resistant. The 'R' signifies splash resistance. . Type I, II, and II R masks are medical masks that are tested according to the exhalation direction (from inside to outside) and take into account the efficiency of bacterial filtration.
EN 14683 2019 has the certain requirement on the construction, design, performance, and testing methods for medical mask that are intended to prevent infectious agents (bacteria, protozoa, viruses, parasitic worms, and fungi) from being transmitted from patients to medical personnel during medical scenario. An appropriate microbial barrier for medical masks can be applied to reduce the transmission of infectious agents from the mouth and nose of an asymptomatic carrier, or a patient suffering from clinical treatment symptoms.
Type I: Bacterial filtering effectiveness > 95%.
Type II: Bacterial filtering effectiveness > 98%.
Type IIR: Bacterial filtering effectiveness > 98%, splash-resistant
![]()
*R = Fluid Resistance
Fluid resistance is the ability of the surgical mask to prevent fluid transfer from outer layers to inner layers as a result of splashes or sprays. EN14683 requires that fluid resistance be tested with synthetic blood at pressures 16kPa. This allows for the testing to determine if the mask is capable of restricting fluid transfer from the outer layers to inner layers as a result of a splash or spray.
All the information contained in this article is for general reference purposes. Kingfa Medical does not guarantee the accuracy, relevance, and timeliness of any information, and the company does not assume any responsibility for errors or omissions in this article.
Reference :
Bissett, J. (2021). COVID-19: A guide to face masks - DentalNursing. Retrieved 26 October 2021, from https://www.dental-nursing.co.uk/news/covid-19-a-guide-to-face-masks




